Keeping your pool water properly balanced is one of the most important tasks a pool owner has, besides throwing cool, summer parties, of course. Most pool owners have a love-hate relationship with pool chemistry. They love it when their water levels are perfect and hate it when they are spending a ton of money to get it balanced again. Balancing your pool water should not be a daunting task. In fact, it could be as easy as A-B-C.
We get tons of calls and emails from customers asking us how to balance their pool water. There’s not one step by step guide that will work for every pool because every pool is very unique. However, if you can understand the relationship between the chemicals in your pool, then it becomes easier to manage your levels.
Balancing your pool water requires an understanding of the relationship between different chemical measurements in your pool. Your pool water’s chemistry is constantly in flux and therefore, chemical levels aren’t the same in January as it is in October. This makes understanding how one chemical affects the other crucial to balancing your pool.
The Basics
The word ‘balanced’, when discussing pool chemistry, comes from the idea that as water dissolves, it holds minerals until it becomes too saturated to hold anymore. Balance is the level at which water is neither over-saturated nor undersaturated.
In order to understand the relationship between each chemical level, you first need to know what you are measuring.

Free Chlorine (FC)
In order to have a balanced swimming pool, you have to maintain the correct levels of free chlorine (FC). Free chlorine is the available amount of chlorine that has disinfecting power. Free chlorine is consumed by two things, namely sunlight and organic materials in your pool. The amount of FC you need to maintain in your swimming pool depends on your cyanuric acid levels.
Check out TroubleFreePool.com’s chlorine/cyanuric chart for guidelines on the appropriate FC levels to maintain based on your cyanuric acid levels.
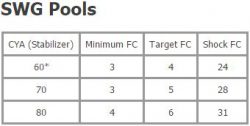
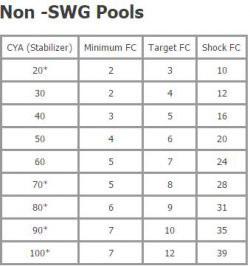
 When your FC is too low, algae are prominent to form. If your FC reaches 0, or you already have an algae bloom, we recommend you do not swim in the water until levels are balanced.
When your FC is too low, algae are prominent to form. If your FC reaches 0, or you already have an algae bloom, we recommend you do not swim in the water until levels are balanced.
Ways to raise FC: household bleach, liquid chlorine, and a salt-chlorine generator.
Acidity (pH)
Your pH level indicates how acidic your pool water is. The pH is a scientific scale from 0-14, with 7 being dead neutral. The ideal pH level is a 7.7 or 7.8. Anything between 7.5 and 7.8 is good, although, anything between 7.2 and 8.0 is acceptable for swimming. Everything that enters your pool, including rainwater, has a pH. So, it’s always good to check your pH levels once a week, to be on the safe side.
 Once your levels fall below 7.2, swimmers tend to experience stinging and burning eyes. Levels below 6.8 can cause severe damage to metal parts in your pool equipment if it is left untreated. It does not take long for a pool with low pH levels to weaken vinyl and heat exchangers.
Once your levels fall below 7.2, swimmers tend to experience stinging and burning eyes. Levels below 6.8 can cause severe damage to metal parts in your pool equipment if it is left untreated. It does not take long for a pool with low pH levels to weaken vinyl and heat exchangers.
Typically, to adjust the pH in your pool, you add pH increasers (bases) or a pH decreaser (acids).
Total Alkalinity (TA)
Alkalinity is closely related to pH. It measures all of the carbonates, bicarbonates, hydroxides, and other alkaline substances that are found in your pool. Total alkalinity is the water’s ability to withstand changes in your pH levels. The pool experts may refer to this as the “buffering capacity.” In a sense, the alkalinity in your pool keeps  your pH levels from fluctuating all over the place.
your pH levels from fluctuating all over the place.
Gunite and concrete pools should keep alkalinity levels between 80-120 ppm.
Painted, vinyl, and fiberglass pools should be maintained between 125-170 ppm.
Low alkalinity can be raised by adding a base, typically sodium bicarbonate(baking soda), while high alkalinity is typically lowered by adding an acid.

Calcium Hardness (CH)
When talking about calcium hardness, you are trying to determine how ‘hard’ or ‘soft’ your water is. ‘Hard’ water has high levels of calcium and magnesium. If these levels are too high, your water becomes too saturated. As a result, these extra deposits looks for other areas to deposit themselves on. Can you guess where they end up? On all types of surfaces in your pool. This includes your pool walls, ladders, lights, and stairs. In fact, you may even see little crystalline clumps all over your pool. Pool experts refer to this as scaling.
Once your CH levels are too low, your pool water is considered undersaturated. If your water becomes undersaturated it starts to look for other sources of calcium like your pool plaster. If it becomes too aggressive, it has the ability to corrode the inside of your pool surfaces.
starts to look for other sources of calcium like your pool plaster. If it becomes too aggressive, it has the ability to corrode the inside of your pool surfaces.
A healthy range to keep your CH is at 220-350 ppm if you do not have a salt chlorine generator. If you do own one, your levels should be maintained between 350-450 ppm.
You can increase your CH with calcium chloride or calcium chloride dihydrate. You can decrease your CH by replacing some of the water in your pool to offset the balance.
Cyanuric Acid (CYA)
Cyanuric acid, sometimes referred to as stabilizer, protects your FC from sunlight and reserves some of the FC for future use. In a sense, it acts as an umbrella for your FC.  The higher your CYA levels are, the more FC you will need to get the same effect. It is essential to monitor your CYA in order to determine what your FC levels should be.
The higher your CYA levels are, the more FC you will need to get the same effect. It is essential to monitor your CYA in order to determine what your FC levels should be.
Without a salt water generator, your CYA levels should stay within 30-50.
With a salt water generator, CYA levels are typically kept between 70-80. You can increase your CYA levels by adding more cyanuric acid.
In most cases, the best way to decrease your CYA levels is simply to replace your water.
Once you have a basic understanding on how each level chemical affects the other, balancing your pool becomes a little less confusing and a lot more manageable. With a lot of things in life, the more you do something, the more comfortable you become with the process.
If you are unsure about your pool’s levels or came up with inaccurate readings, have your water retested. Many local pool stores tests pool water for free. And if you are still unsure, have it tested again. There is no harm in testing your water frequently.
**Acknowledgment: The original Chlorine/CYA “Best Guess” chart was developed by Ben Powell. Richard Falk (chem geek) has refined and expanded on that original to produce theChlorine/CYA Chart by Chem Geek based on experiences with members on Troublefreepool.com and the old Pool Forum and conformation to chemical theory**












We can’t get a chlorine reading in our pool is no how much we shock it. The readings are:
FC – 0.48
TC – 0.48
PH – 7.6
Alkalinity – 91 (adjusted due to the effect of CYA on tested total alkalinity)
CH – 380
CYA – 74
Iron, Copper and Phosphates – 0
TDS – 2200
Your water looks relatively balanced; the only point of concern I see is the TDS levels. Anything above the 2,000 mark is not ideal; you’ll need to drain and refill with fresh water to get that number down.
We have a 24 round pool, changed liner this year so it has fresh water but alkalinity level is high and no chlorine registering!
We have an inground pool. We started having difficulty last year with the alkalinity being very high 22+ (220+). Scale built up on the sides and such so we drained the pool and cleaned the best we could. After refilling the pool, the PH is running around 7.2 to 7.6 but the alkalinity is still over 20. It is holding chlorine well at this point. So do I add acid to drop alkalinity and then add ash to bring up PH, but won’t that also bring up alkalinity again? I’m stumped.
We have a 25’ above ground pool. Our water has remained cloudy all season. Our small kit showed we had no chlorine no matter how much we added. We have shocked & used clarifier, vacuumed it almost every day. We just got a new test kit that showed free chlorine was 2-4, no combined chlorine, cya 65, ph 7.1 (first time ph has been out of whack all summer) Please help! I’d like to be able to see my feet in the pool.
I just installed a 24′ round salt water pool with chlorine generator. Filled it up with well water – took a few days so while it was filling up I would throw in every once in a while 4 litres of water with about 4 crushed stabalized chlorine tablets – because it was going green. Then when it was full I added 6 or 7 20kg bags of salt to bring it up to the proper salt level (it’s sitting around 3100 ppm). Initially the pH level was ok until I added the salt which then brought the highest reading on the test strips I think 8.6 and the Alkalinity was very high as well 240ppm (the highest on the test strips). Chlorine reading was zero. The stabalizer was reading low as well.
So the instructions as I read off the chart with the pool said I need to bring the pH down with Muriatic acid and the chart said 10Litres of the stuff. I didn’t believe it, so I just got 2 – 4L jugs grade 20Buame 31.45% and used one entire jug into the pool, it was some pretty powerful stuff. I indeed got the pH down but it went down to almost 6 pH so I quickly ran to the store and got some pH+ booster (powder kind) – I didn’t use more than a cup but I think I still need to bring the pH up – it’s around 7 or so.
My question is did I use too much muriatic acid? I think I did. Does it dissipate over time? Should I use more pH+ in the pool? I was thinking I should backwash a little to remove some of the chemicals and put fresh water in to dilute the chemical mess I might have created. It has since rained a few times so that added new clean water to the pool.
I’m always leery about adding acids to any clean water, Is there anything bad about the pH+ cyuranic acid. I know the muriatic acid is bad (after adding the 4 litres) I guess the pH was still dropping and I added a bit more but not much. Anyways we’ve had a few swims in the water already and it doesn’t seem too bad. pH I think is in the 7 range now but the strips only give you a rough kind of reading. Should I purchase a digital meter tester?
I guess the pool will get diluted out the more it rains but I was quite concerned with the muriatic acid when I dropped the pH to 6 or so.
We just put in a new liner in our inground 20*40 pool and used the stream to fill it. We put in 4 gallons of shock and tabs in the strainer, but CL Free and total is 0, ph is lower than 7 and alkalinity isn’t on the chart. I just added stabilizer. What next? It isn’t that warm, but it is sunny.
This should be pretty straight forward: add pH increaser, alkalinity increaser, and check heck you stabilizer levels to determine how much needs to be added.
Our above ground 16× 32 pool won’t hold chlorine. We have tried everything. We have had our water tested, but they haven’t been any help.
In order to balance your water, I’d first have to know what is imbalanced. Post the results of your last water test.
How do you raise alkalinity but not cause PH to raise also. My alkalinity was low but my PH was 7.6. When I tried to fix A it made PH go higher. So I just kept bouncing back and forth.
Chris – the easiest way to raise your alkalinity with less of an affect on your pH is by adding baking soda. Feel free to use the TroubleFree Pool Math CalculatorTroubleFree Pool Math Calculator.
Do you think you can help us with this. We have spent a lot of money so far and are still in the same situation
Help, our pool has been cloudy for a few months now. We had it tested and was told to put in 4packs of powder, powder (clorene) four days in a row. Nothing happened, the water is still cloudy, we even put some clarity in. The pool is in the ground, 33,000 gal. (approx.) and is supposed to be salt water. The p/h level is said to be fine, but were still in a quandry. Can you help or give us some advice?
Gina, we can try to help you. First, can you post the results of your most recent water test? We need your test results to determine where we need to start.
The info we need:
Free Chlorine (FC) =
Combined Chlorine (CC) =
ph =
Total Alkalinity (TA) =
Calcium Hardness (CH) =
Cyanuric Acid (CYA) =
Gina – I hope by now you have figured out your problem. I had the same problem also but my pool is above ground and I use (now) a sand filter. I found that the best thing I did to get a crystal clear pool was to use plain ole bleach instead of the powder shock. I noticed that when the powder was mixed in the bucket that it was cloudy too. So I have gotten away from using powders. I also used Robarb Super Blue Cloudy Pool Clarifier. It is a liquid and not one of those “flocks” that take the stuff to the bottom. First day my pool was bright blue and then two days later it was crystal clear! I also ran the filter 24/7 until it was clear and backwashed it 3-4 times a day. Hope this helps.
You have mentioned pretty much of those things I’m aware of but, I’m dealing with a problem I never heard of before and certainly never expected. I have been unable to maintain the right level of chlorine even a day after adding a full gallon in a 17,000 gallon pool. I’m told the reason is my phosphate level is up around 1000. What is going on? Are you aware of this condition? Do others have this problem? Could use some help here!
Can you post the latest results of your water test? The list should include Free Chlorine, pH, Total Alkalinity, Hardness, and CYA. This will give a better idea of what’s the matter.
“Low alkalinity can be raised by adding a base, typically sodium chloride (baking soda), while high” Should be sodium bicarbonate, baking soda is sodium bicarbonate,not sodium chloride aka salt.
Thanks for the catch. We have edited the article.