We all know that chlorine is necessary to keep our pool sanitized, but do you know the relationship it has with cyanuric acid? Chlorine and cyanuric acid help fight germs, bacteria, and other viruses that may enter our pool at any given time. In fact, chlorine kills bacteria with a 99.9% reduction in less than a minute. Still, there are a ton of questions about chlorine that pool owners still grapple with. Is there such a thing as having too much chlorine? At what point does your pool water become unsafe to swim in due to over-chlorination? How much chlorine do I add to my pool? Is it also true that chlorine is a carcinogen? These questions have multi-faceted answers, so let’s dive right in.

Free Chlorine, Combined Chlorine, and Total Chlorine
First, let’s make sure you understand the differences between free chlorine, combined chlorine, and total chlorine and how you measure each.
Free chlorine (FC) shows the level of disinfecting chlorine available to sanitize your pool. FC is the part of the chlorinated water that hasn’t interacted with contaminants, yet.
Once chlorine interacts with compounds such as urine, sweat, bacteria, etc they form chloramines. Chloramines are what we refer to as combined chlorine (CC). The CC levels are chlorine that no longer has disinfecting power. Essentially, it is useless.
Total chlorine (TC) is simply your FC plus your CC. Your TC should not be less than the FC level.
The Chlorine and Cyanuric Acid Relationship
Though the relationship between chlorine and cyanuric acid is not a new revelation to the pool industry, it is very rare that you come across pool technicians who truly understand the effect one has on the other.
Before you can recognize when you have too much chlorine in your pool, or not enough, you have to know the recommended levels your pool needs. And you won’t be able to understand the recommended levels until you understand the relationship chlorine has with cyanuric acid (CYA) and pH.
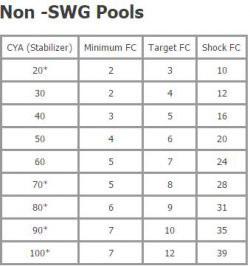
You might have read somewhere that the amount of chlorine you need to add to your pool depends on your current CYA levels . Remember, FC not only gets depleted through interacting with germs and bacteria but is also consumed by sunlight. It has been stated that 90% of unprotected chlorine is lost within two hours by direct exposure to sunlight. In a sense, CYA acts as a reservoir for FC. So, if you live in a state like we do (Florida), it’s something you have to consider.
It is essential to maintain the FC/CYA ratio to kill algae and bacteria effectively. It’s been proven that by keeping the correct CYA/FC ratio, you can control algae growth under all conditions. Given a minimum FC concentration, the FC/CYA ratio determines algal growth rates. It is recommended that your FC is 7.5% of your CYA levels. This ratio determines the active chlorine levels needed to prevent algal growth, not just FC alone.
For example, if your pool started with 30 ppm of CYA, then 7.5% of 30 ppm is 2.25pm of FC. As your CYA increases, higher levels of FC are needed in order to prevent algal growth. Below is a chart we pulled from TroublefreePool.Com showing the recommended relation for outdoor residential pools.
Now that we know how CYA affects chlorine, let’s add pH to the mix.

Adding pH to the Mix
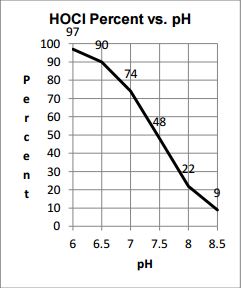
In relation to chlorine, the pH of your pool water determines how effective your chlorine is. Higher levels of pH threaten the disinfecting power of chlorine.
To the left is a chart showing the effectiveness of chlorine in relation to pH values when your CYA is zero. You can see that without CYA in your pool and high pH levels, your chlorine becomes ineffective extremely fast. At that point, it doesn’t matter how much FC is available because it’s probably not doing the job.
The pH scale ranges from 0-14, where 7 is considered neutral. Ideally, it is recommended to keep your pH around 7.4, the same as the pH in the human eye. This also gives chlorine good disinfecting power.
Common Chlorine Myths
After helping pool owners for a few years, you realize that there is a ton of bad information on the Internet . Not just bad information, but completely incorrect information. So here are a few myths about chlorine that you may or may not know.
You can smell when there’s too much chlorine in the pool
False. If your pool guy tells you this, politely take a step back, and go in the opposite direction. A properly sanitized pool will not have a strong chemical smell. The strong smell common to many pools, especially public pools, is chloramines. Chloramines are what’s left once chlorine interacts with all the contaminants (i.e. germs, bacteria, soap, dirt, etc.) in the pool. In reality, the chemical smell you think is chlorine is not an indication of a clean pool, but rather a sign that you may need to check your chlorine levels. At this point, the chlorine is working overtime to eliminate all the contaminants in the pool and chlorine may need to be added.
Too much chlorine in the pool stings your eyes.
It’s actually just the opposite. Your eyes are stinging because there’s probably not enough chlorine in your pool. According to the CDC, “Chloramines in the water, like dichloramine and trichloramine, irritate skin, eyes, and the respiratory tract…” As mentioned earlier, chloramines are the byproducts formed when chlorine interacts with all the nasties in your pool water. For red, stinging eyes, the culprit is typically urine. Urine binds with chlorine to form a specific chloramine, which in turn, causes your eyes to sting and turn red. To be specific, it’s not the actual urine, it is the ammonium found in urine. So, the next time someone complains that his/her eyes are red or stinging, you will know it’s because someone peed in the pool.
Too much chlorine caused my daughter’s hair to turn green.
This is another widely spread, misconceived notion pool owners have. Chlorine does not directly turn your hair green nor is chlorine green itself. This happens when there are large compounds of metals, predominantly copper, in the water. The copper interacts with the chlorine in the water to form a compound that binds to your hair. The cuticle, which is the outer covering of your hair, breaks down after it is exposed to high levels of copper. Once in your hair, it oxidizes and turns green. (Think of the color of the Statue of Liberty.)
Although chlorine does not cause your hair to turn green, it is the chlorine that caused the copper to stick to your hair.
Chlorine is considered a carcinogen.
Despite numerous websites, web doctors, and self-proclaimed scientists, there is no current evidence that proves drinking and swimming in water with chlorine causes cancer. So, stop reading the comments section in these articles. Yes, chlorine and chlorine emissions can aggravate respiratory conditions and other health complications, however, there is still no foundation or study that links cancer to chlorine.











So, what’s the verdict everyone? Liquid Chlorine?? My CYA levels are 166. We are fairly new to the pool scene abc had no idea about CYA. The pool has always been at nearly 100% since we’ve lived her. But all of a sudden, BAM! We drained pool 2 feet and took out the Trichlor tablets. Shocked it and two days later CYA is practically the samPlease help!! The pool is gorgeous, it’s May in Texas, over 90* outside and we can’t swim because fear of unsanitary!??
Drain more water and fill until your cya comes down to correct levels
I just drained and refilled my pool two weeks ago, as my CYA was at 165.
I didn’t want to drain the whole pool, but once I thought about it, I realized it was the only logical option.
If I drained 50% and refilled with tap water (containing 0 CYA), my CYA would only come down to about 88ppm – still above the ideal range.
If I drained 75% it could have got down to 42ppm, but once you’ve drained 75% of your pool and knowing it’s old water, I felt like at that point it just made sense to put in all new water.
I’ve been battling with rebalancing. This is my first pool and have only been doing the chemistry for a couple years. I’m finally about there. Just had my water tested and I’m at 2.54 FC, 7.6 pH, 65 Total Alk, 226 Cal Hardness & 26 CYA.
I’m on the fence if I should add a smidge of conditioner to bring the CYA up over 30 or just let my Trichlor tabs do the work.
I might risk it and let the stabilizer in the trichlor tablets get it over the 30 ppm goalline. You’re pretty close, so the stabilizer should accrue relatively quickly.
i’d never but trichlor or conditioner and or dichlor or anything else with cyanuric acid in my pool.
Good article !!! I have a 40,000 gal. liner, deep in the trees. Always beautiful thanks to dad. A trick my father used to counter the effect of high CYA was to keep the FC at 7% of CYA and let the lawn sprinkler spritz the surface of the pool each night for 15 mins. it replenishes the water slightly, but more importantly, the water dilutes CYA at the surface, increasing FC at the surface and use only non-stabilized chlorine until CYA is down. I’m on a well, so i use calcium based chlor. to keep the calcium up. no draining, no overfill.
No need to keep calcium up if you have a vinyl liner pool.
Is there a level at which there is _too_ much CYA? If so, what are the consequences of it?
Too High of a CYA level (above 70) will require you to keep your free chlorine levels higher to be effective. This gets impractical and expensive. It will also require you to use way more chlorine in the event you need to shock/slam. Personally, i keep my CYA around 40 and Only use tablets when going on vacation as they contain CYA stabilizer in their ingredients.
How do you keep your chlorine level without adding tablets? only using shock?
I didn’t use tablets during winter to reduce the CYA and even doing that did not go down much. Currently is on 79. Last summer was a nightmare to keep the Chlorine and Algaes away. I was hoping for better this coming summer. However, looking that my CYA still high I don’t know what else to do before the actual summer begins. Please help!!
Just to get yourself some Liquid Chlorine, otherwise known as Bleach. You can use the 6% common bleach (only use the stuff that has no additives or perfumes, etc). Or you can get higher concentrations 10% or 12% from Walmart or Lowes or other big box stores.
Just as an example, I have a 36,00 gallon pool. In order for me to raise the Chlorine from say 1ppm to 3 ppm, would ad 92oz of 10% liquid chlorine (bleach). The nice part is that it’s just chlorine that’s being added. No CYA, no Calcium, etc.
Now, if someones CYA is low, they could in theory use trichlor or dichlor for a while to build it up.
Great info I think I learned a lot but I’m still unsure about my pool. I used pool tablets for a long time thinking everything was fine I then started putting in stabilizer to make the water crystal clear. Wich it did. I didn’t use the pool over the winter just kept tablets in it. Then as soon as spring hit this year pollen dropped in the pool and started to stain the floor. I scrubbed and cleaned it every day but couldn’t keep up. The yellow staining soon turned to algae. I checked the levels and found my cyanic acid was really high 150ppm. So I added flocculant and then vacuumed up the debris. Stoped using tablets and actually had to drain the pool almost completely twice within two weeks. So after all that my cyanic acid is still the same 150ppm I’m able to keep all the other levels at a there normal safe zones but can’t control or even change the cynaric acid at all. I am adding a lot of chlorine as well to keep it good about a half a jug per day for 1600 gallons. Any thoughts?
Check if the chlorine or pool shock you’re adding has CYA. I’ve found the shock and #-in-1 chlorine mixes all contain CYA components (ie. Trichloroisocyanuric Acid). Only the gallon jugs of sodium hypochlorite would be cya free.
There’s a few things to unpack here. Stabilizer does not make the water crystal clear, it buffers the chlorine so it can do its job before burning off from the sun. But you say you’re using tablets.
If you’re using tablets, you’re adding CYA every time. This means you need more and more chlorine to be effective,to the point of being impractical. 150ppm is way too high! At 150cya, you’d need an FC of 18ppm to be effective! That would be really hard to maintain (and expensive). Unfortunately (or fortunately) cya does not evaporate, so you’d need to drain water and replace it to lower that concentration. Stop using those pucks, they are causing the problem!
It’s important to understand that cyanuric acid also can easily give false free chlorine readings. You can have zero FC and OTO a reading of 3 ppm.
If you want a truly accurate FC reading, you’ll need a DPD test
I’m struggling with a cloudy pool. Actually you would define it as opaque. My Ph and hardness levels are within the recommended numbers. My CYA level is negligible and so was my chlorine. Can you give me some pointers? What do I need to do? I have an 18’ x 48” round Coleman above ground pool. It has a cartridge filter. I’ve shocked and added clarifier a couple of times and I skim the water with a skimmer net. I continuously clean the cartridge (3 or more times a day). I tried to vacuum, but I can’t see the bottom to know where the debris is.
Have you added any algaecide that contains copper?
So, for a CYA level of 25
I need at least about 2 ppm of Chlorine
But if the PH is at about 7.8, on which only 1/3 of the chlorine will work, than I need a chlorine level of about 6 ppm, right?
No. Just lower your PH level
Why does my lazy river fc drop during the day slowly
Did you get a chance to read the article? What are your water chemistry levels?
Hey Matthew my concern is my 4 year old sometimes gets to throwing up a day after being in our pool.
Pretty much he just stays in the 90 degree jacuzzi, swims underwater a lot, and he loves to squirt water out of his mouth. 26,000 Gallon in ground pool. Cycles water at least once a day through Pentium Cartridges. I really like minimal chlorine. I have not been shocking with SCHOCK but using liquid chlorine instead. I also use Pool RX in Spring and Summer to help with green algae.
Pool RX has copper. Can you name a Sanitizer product or chlorine that has no copper.
Total Alkalinity is 82. Ph is 7.9 , Chlorine is 1 and Free Chlorine is .5 CYA is 90.
Pool water is very clear and clean to the eye.
“can you name a sanitizer without copper” yes. Liquid Chlorine. CL has no copper.
“I really like minimal chlorine”. Then you haven’t understood the article. If you had, you’d know chlorine is not the bad guy. Stop and ask yourself why you “like minimal chlorine”. The answer is because, like many people, you’ve learned the common myths. Have a read through this article again, or check out some of the chemistry basics on INYO or Troublefreepool to learn enough to dispel those myths. You don’t need poolrx and you don’t need shock.
Ps. I’m not a doctor, but I’d say your 4yr old might be throwing up from being in the jacuzzi. Hot tubs aren’t meant for young kids who can overheat really quickly. Can you get heat stroke from a hot tub? You bet you can.
Could it be that your kid is throwing up because he is drinking a bunch of pool water?
Really amazing info
Good info. Thanks.
This is the perfect way to break down this intnomafiro.